argon
A QUICK GUIDE

Atomic Number: 18
Atomic Weight: 39.948
Melting Point: 83.80 K (-189.35°C)
Boiling Point: 87.30 K (-185.85°C)
Phase at Room Temperature: Gas
Element Classification: Non-metal
Period Number: 3 Group Number: 18 Group Name: Noble Gas
Argon is a colourless and odourless gas which can be found in the atmosphere. It is a non-metal, and part of the group of the noble gases. It cannot be found in any compound forms.
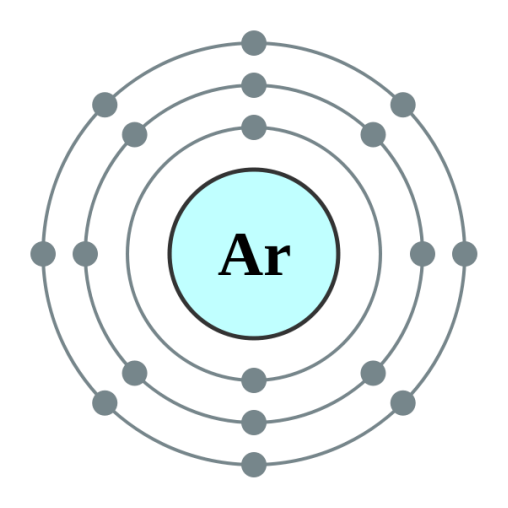
Electron Configuration
As Argon has an electron configuration of 18, all of the orbitals are filled with electrons and therefore do not need to give away any electrons, making it a very stable element. It does not produce any stable compounds at room temperature.
Atomic Weight: 39.948
Melting Point: 83.80 K (-189.35°C)
Boiling Point: 87.30 K (-185.85°C)
Phase at Room Temperature: Gas
Element Classification: Non-metal
Period Number: 3 Group Number: 18 Group Name: Noble Gas
Argon is a colourless and odourless gas which can be found in the atmosphere. It is a non-metal, and part of the group of the noble gases. It cannot be found in any compound forms.
Electron Configuration
As Argon has an electron configuration of 18, all of the orbitals are filled with electrons and therefore do not need to give away any electrons, making it a very stable element. It does not produce any stable compounds at room temperature.
Pictured above: A Bohr model of the electron configuration of Argon.
In the Environment
Argon gas occurs naturally in the environment. It it not known to harm any being, and is part of the Earth's atmosphere. Argon makes up 0.93% of the earth's atmosphere, making it the third most abundant gas and has the same solubility of oxygen
Uses
Argon in its gaseous form is commonly used when a stable gas is needed. It is used in the production of titanium and other reactive elements as a sufficiently inert gas. Argon is used in fluorescent tubes and low-energy light bulbs. A low-energy light bulb often contains argon gas and mercury.
Argon gas occurs naturally in the environment. It it not known to harm any being, and is part of the Earth's atmosphere. Argon makes up 0.93% of the earth's atmosphere, making it the third most abundant gas and has the same solubility of oxygen
Uses
Argon in its gaseous form is commonly used when a stable gas is needed. It is used in the production of titanium and other reactive elements as a sufficiently inert gas. Argon is used in fluorescent tubes and low-energy light bulbs. A low-energy light bulb often contains argon gas and mercury.